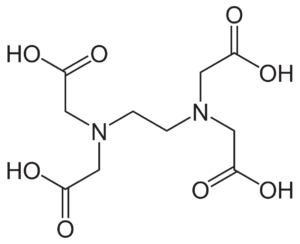
Tetrasodium EDTA is a water-soluble ingredient.
Uses
- Used as a chelating agent in cosmetics and personal care products.
Benefits
- It has the ability to sequester metal ions and allow them to remain in formulas but lose their ability to react with other ingredients. This ability allows it to not only soften water, but preserve formulas as well by improving its stability when it is exposed to air and prevent microbial growth
Cautions
- The Cosmetics Database considers Tetrasodium EDTA a low to moderate hazard ingredient, depending on its usage. It notes concerns regarding cancer, enhanced skin absorption, organ system toxicity and irritation (due to animal studies that showed skin irritation at low doses). It is classified as a high human health priority and expected to be toxic or harmful.
- However, tests done by the CIR (which are not corroborated) found that Tetrasodium EDTA is not a dermal irritant or a sensitizer, and is not absorbed by skin easily. “Because these ingredients bind metals required for normal cell division, some studies that indicated that these compounds were weakly mutagenic. Some studies showed reproductive and developmental effects following oral exposure to large doses of metal chelators, likely an effect of the binding of metals required for normal reproduction and development” (RealSelf.com). It found that cosmetics and personal care products containing these ingredients would result in very little skin penetration and systemic levels well below those shown to produce adverse effects in oral studies.
- Tetrasodium EDTA has been found to be an ecotoxin, however. “Widespread use of EDTA and its slow removal under many environmental conditions has led to its status as the most abundant anthropogenic compound in many European surface waters … and has emerged as a persistent organic pollutant” (Wikipedia).
Interactions
n/a
Other names
n/a
References
Source: TruthinAging, https://www.truthinaging.com/ingredients/tetrasodium-edta

Soothes irritated skin such as Eczema, Psoriasis, Burns, Dry-Cracked Skin, Bug Bites, Sunburns, etc.The combination of natural and organic ingredients have beneficial properties for skin.
Helps to reduce the look of fine lines and wrinkles. Makes your skin feel softer and smoother. Hypo allergenic and safe. Doesn’t contain harmful chemicals. Good for all skin types, especially sensitive skin. Visit the website at http://deluviausa.com/product-category/deluvia-skin-care/
Click to View what Alicia W., Co-Founder of Delúvia Inc. has to say about how the company started and how its products could help others.
https://www.youtube.com/watch?v=ePrifdk1Wus